How is Chlorine made?
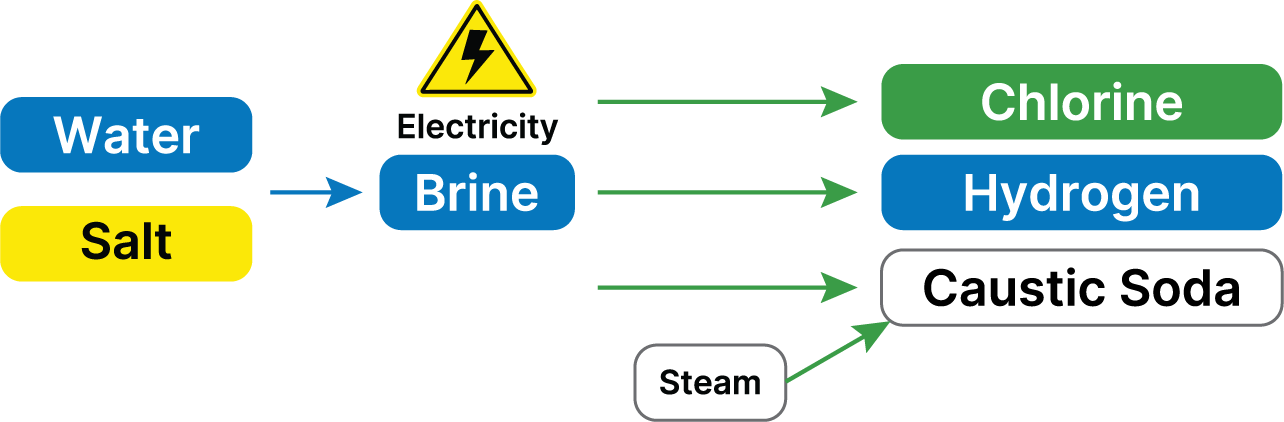
Chlorine is made in a process known as electrolysis which also produces Sodium Hydroxide (Caustic Soda) and Hydrogen as co-products. Electrolysis is a chemical process which uses electricity to split salt and water into Chlorine, Caustic and Hydrogen. This takes place in an electrolyser. An electrolyser is made up of a positive electrode (or anode) and a negative electrode (or cathode). These electrodes are sat in salt water with the two electrodes being separated by a semi-permeable ‘barrier’ (also known as a membrane or a diaphragm). This barrier keeps the Chlorine safely separate from the Caustic and Hydrogen.
Europe makes around 8 million tonnes of Chlorine each year and you can find out more about European production of Chlor-Alkali here.