Find all documents and resources here
News & Press Releases
Read our news, press releases and monthly production statistics below

03 Apr 2026
February 2026 Chlorine production

02 Apr 2026
Euro Chlor at the Chlorine Institute 2026 Annual Meeting: technical, safety and regulatory insights

01 Apr 2026
January 2026 Chlorine production
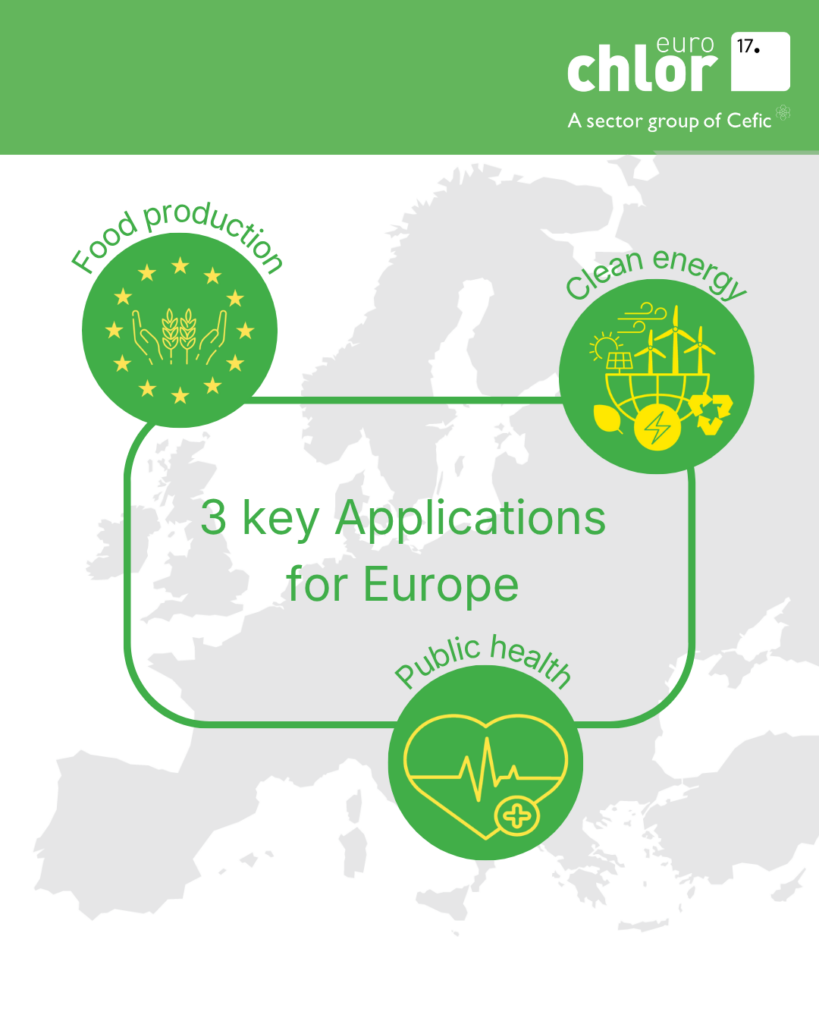
01 Apr 2026
Why Europe needs local production of Chlorine

20 Feb 2026
December 2025 Chlorine production

20 Feb 2026
November 2025 Chlorine production

18 Feb 2026
October 2025 Chlorine production

24 Nov 2025
Ting Ting from BASF features as fourteenth 17 Chlor-Alkali Careers interviewee

07 Nov 2025
Marcel from VESTOLIT features as thirteenth 17 Chlor-Alkali Careers interviewee

04 Nov 2025
September 2025 Chlorine production

02 Oct 2025
August 2025 Chlorine production

23 Sep 2025
New Chair welcomed for Euro Chlor Statistics Committee

17 Sep 2025
New Euro Chlor Manager announced at 2025 General Assembly

15 Sep 2025
Euro Chlor appoints new Management Committee Chair

10 Sep 2025
Euro Chlor launches Chlor-Alkali Industry Review 2024-2025

04 Sep 2025
July 2025 Chlorine production

04 Aug 2025
June 2025 Chlorine production

02 Jul 2025
May 2025 Chlorine production

04 Jun 2025
April 2025 Chlorine production

15 May 2025
Technology Conference stays strong and concludes with focus on Safety, Sustainability and Future challenges
